| Basic
Information |
| Name |
Lithium
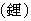
|
| Symbol |
Li |
| Atomic
Number |
3 |
| Atomic
Mass |
6.941
amu |
| Melting
Point |
180.54
°C |
| Boiling
Point |
1347.0
°C |
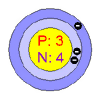
| Number
of Protons/Electrons |
3 |
| Number
of Neutrons |
4 |
| Classification |
Alkali
Metal |
| Crystal
Structure |
Cubic |
| Density@293
K |
0.53 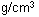 |
| Color |
Silvery |
|
| Number
of Energy Levels |
2 |
| First
Energy Level |
2 |
| Second
Energy Level |
1 |
|
 |
| Isotope |
Half
Life |
| Li-6 |
Stable |
| Li-7 |
Stable |
|
| Date
of Discovery |
1817 |
| Discoverer |
Johann
Arfvedson |
| Name
of Origin |
From
the Greek word lithos (stone) |
| Uses |
Batteries,
ceramics, lubricants |
| Obtained
From |
Passing
electric charge through melted lithium chloride, spodumeme |
|
|